Lorem ipsum dolor sit amet, consectetur adipiscing elit. Ut elit tellus, luctus nec ullamcorper mattis, pulvinar dapibus leo.
Pentose Phosphate Pathway
MCAT Biochemistry Chapter 5 - Section 1.3 - Metabolic Pathways - Pentose Phosphate Pathway
Sample MCAT Question
One of the products of the pentose phosphate pathway, NADPH, is essential for all of the following EXCEPT:
a) fatty acid synthesis
b) oxidation of cholesterol in the liver
c) steroid hormone synthesis
d) reducing glutathione in the body
B is correct. oxidation of cholesterol in the liver.
The pentose phosphate pathway produces NADPH from the electron acceptor NADP+. NADPH is essential for fatty acid synthesis, cholesterol and steroid hormone synthesis, and preventing oxidative damage from reactive oxygen species. It is not used in the oxidation of cholesterol in the liver.
Pentose Phosphate Pathway
Aside from glycolysis, another way that the body can oxidize glucose for use in the cells is through the pentose phosphate pathway. Like glycolysis, the pentose phosphate pathway occurs exclusively in the cytoplasm of cells. But, unlike glycolysis, NADP+ serves as the electron acceptor and is reduced to NADPH.
Note that NADP+ and NADPH are not the same as NAD+ and NADH. These two pairs of molecules actually serve opposite functions in the cell. Recall that NAD+ is used to oxidize glucose to release energy. This is because, within the cell, NAD+ is at high concentrations and NADH is at low concentrations. This concentration difference favors the reduction of NAD+ into NADH, providing the cell the power to oxidize other compounds. On the other hand, the relative cellular concentrations of NADP+ and NADPH provide reducing power to the cell. In the cell, NADPH is at high concentrations, and NADP+ is at low concentrations. This concentration difference favors the oxidation of NADPH into NADP+, allowing the cell to reduce other compounds. This reduction capacity of the cell is useful for a variety of anabolic processes, for example, fatty acid synthesis, cholesterol synthesis, and steroid hormone synthesis, as well as preventing oxidative damage from reactive oxygen species.
Another product of the pentose phosphate pathway is ribose 5-phosphate, which is where the pathway gets its name from. Ribose 5-phosphate is a pentose phosphate, consisting of the 5-carbon sugar, ribose, an essential component for nucleotides and nucleic acids. In other words, the pentose phosphate pathway also produces a vital precursor of DNA, RNA, and other coenzymes such as NADH, FADH2, and coenzyme A.
There are two phases of the pentose phosphate pathway – the irreversible oxidative phase and the reversible nonoxidative phase.
Pentose Phosphate Pathway: Oxidative Phase
The overall reaction of the oxidative phase of the pentose phosphate pathway is shown below:
Glucose 6-phosphate + 2 NADP+ + H2O → Ribulose 5-phosphate + 2 NADPH + 2 H+ + CO2
In the first step of the oxidative phase, glucose 6-phosphate is oxidized to 6-phosphoglucono-Δ-lactone, which is a lactone or cyclic ester (Figure 1). Because this is a redox reaction, while glucose 6-phosphate is being oxidized, another molecule must also be reduced. Here, NADP+ is reduced to NADPH. This overall reaction is catalyzed by the enzyme glucose-6-phosphate dehydrogenase.
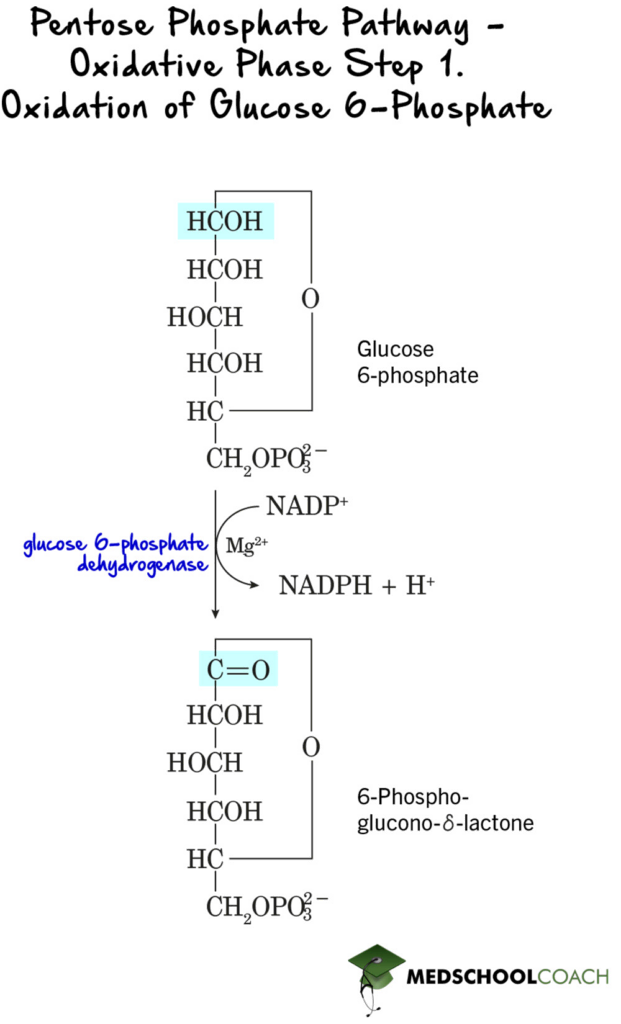
In the second step of the oxidative phase, 6-phosphoglucono-Δ-lactone is hydrolyzed to 6-phosphogluconate by the enzyme lactase (Figure 2). This is a hydrolysis reaction, meaning that water is used to break one or more chemical bonds. In this step, water is used to break apart the cyclic pentose ring.
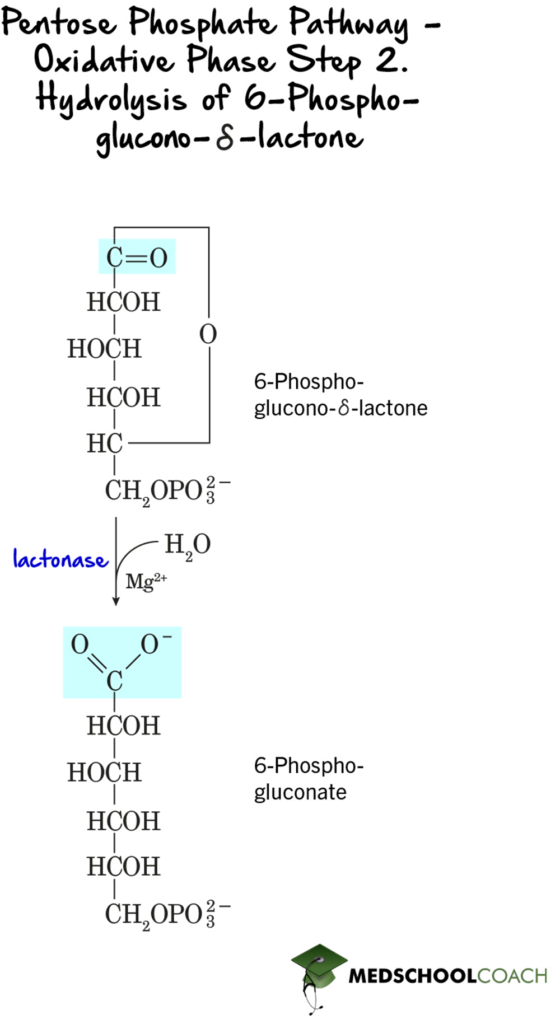
In the third step of the oxidative phase, 6-phosphogluconate is oxidized and decarboxylated to form ribulose 5-phosphate (Figure 3). Again, this step is a redox reaction, and NADP+ is once again reduced to NADPH. This reaction is catalyzed by 6-phosphogluconate dehydrogenase.
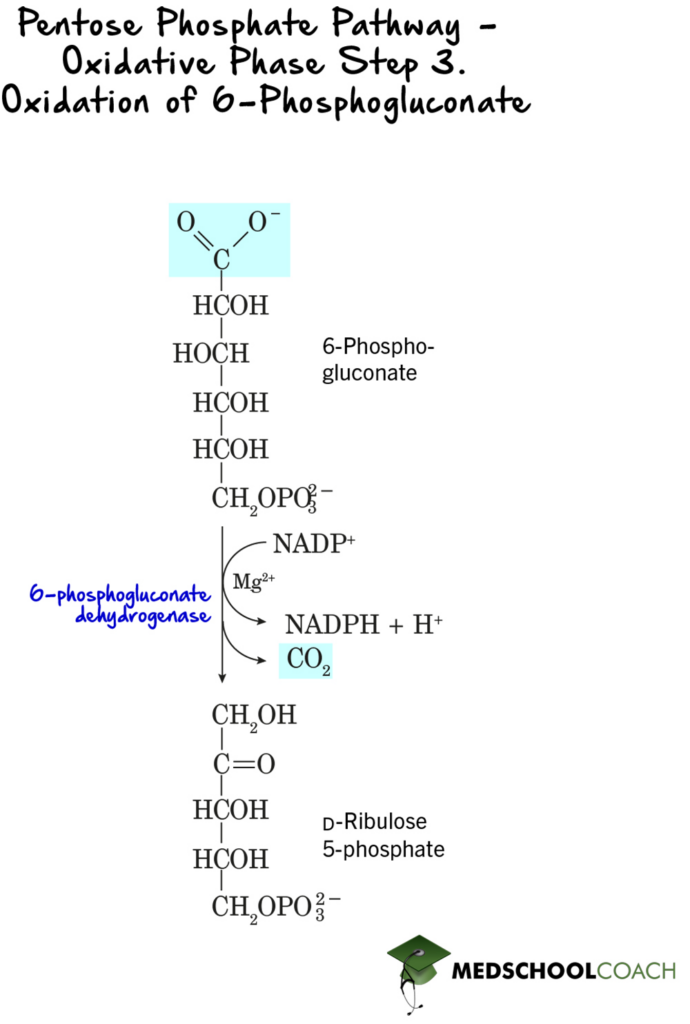
In the last step of the oxidative phase, ribulose 5-phosphate is converted to ribose 5-phosphate by the enzyme phosphopentose isomerase (Figure 4).
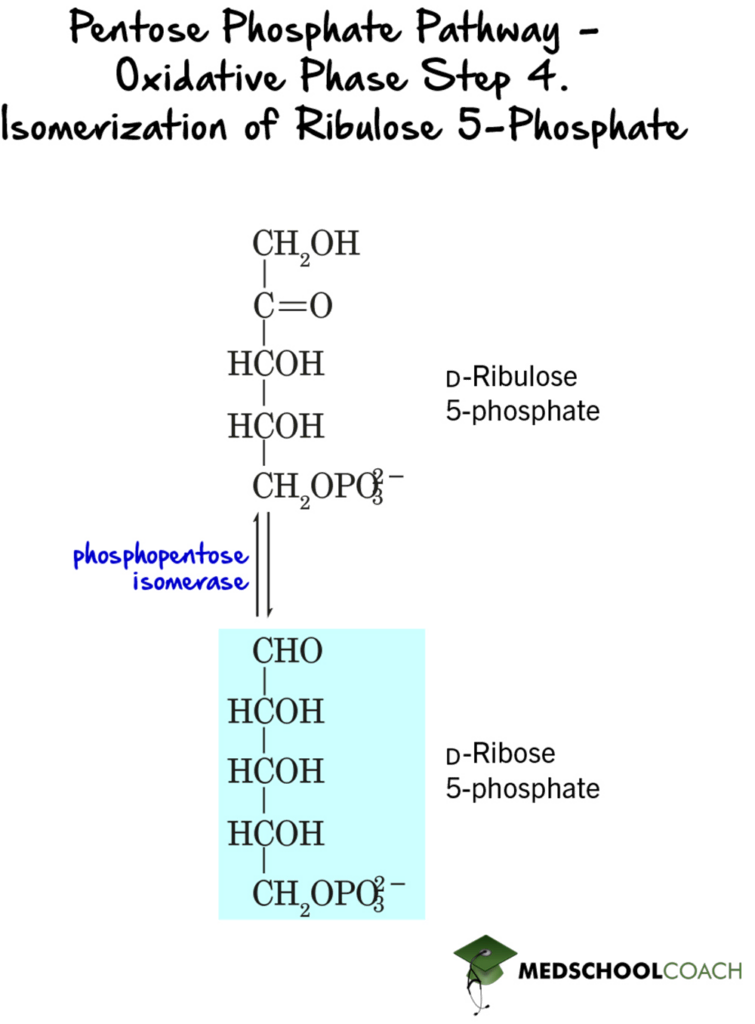
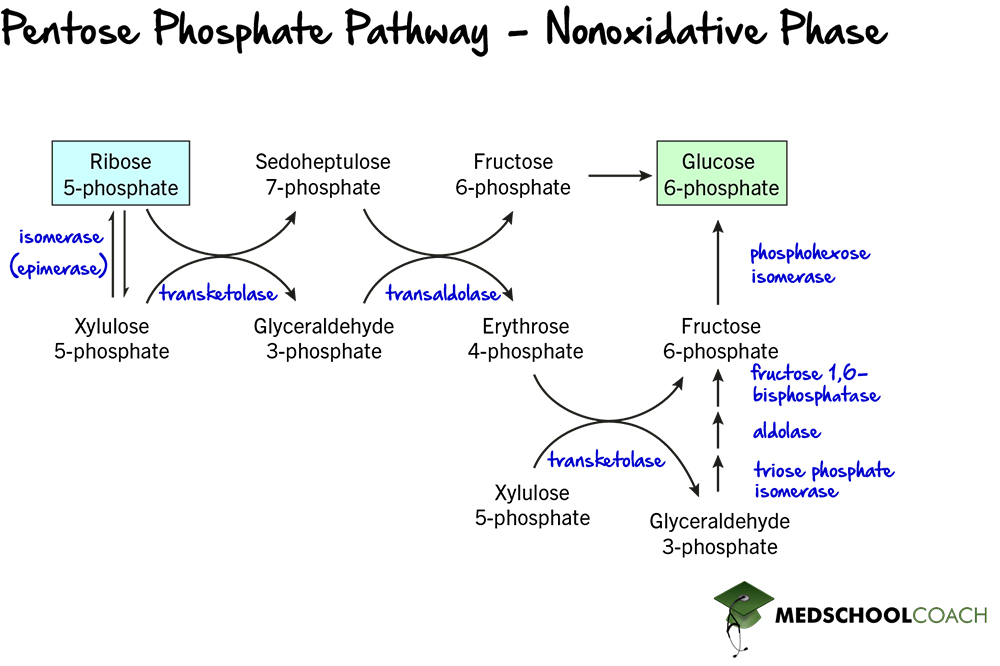
Nonoxidative Phase of the Pentose Phosphate Pathway
The rest of the pentose phosphate pathway steps are part of the nonoxidative phase. This phase consists of a series of reversible reactions that serve to convert ribose 5-phosphate into glycolytic intermediates. Figure 5 shows several different reactions of ribose 5-phosphate. The reversibility of this pathway allows the cell to choose, based on its needs, whether to run the pentose phosphate pathway to produce ribose 5-phosphate or NADPH, or to favor intermediates of glycolysis.
Looking to review additional metabolic pathways? Check out our posts on fermentation, gluconeogenesis, and the regulation of glycolysis and gluconeogenesis!
Explore More MCAT Masterclass Chapters
Take a closer look at our entire MCAT Masterclass or explore our Biochemistry lessons below.

One-on-One Tutoring
Are you ready to take your MCAT performance to a whole new level? Work with our 99th-percentile MCAT tutors to boost your score by 12 points or more!
See if MCAT Tutoring can help me
Talk to our enrollment team about MCAT Tutoring

MCAT Go Audio Course
Engaging audio learning to take your MCAT learning on the go, any time, any where. You'll be on the way to a higher MCAT score no matter where you are. Listen to over 200+ lessons.

MCAT Practice Exams
Practice makes perfect! Our mock exams coupled with thorough explanations and in-depth analytics help students understand exactly where they stand.

MCAT Prep App
Access hundreds of MCAT videos to help you study and raise your exam score. Augment your learning with expert-created flashcards and a question banks.