The Krebs Cycle
MCAT Biochemistry - Chapter 6 - Section 2.2 - Cellular Respiration - The Krebs Cycle
- Home
- »
- MCAT Masterclass
- »
- Biological and Biochemical Foundations of Living Systems
- »
- Biochemistry
- »
- The Krebs Cycle – MCAT Biochemistry
Sample MCAT Question - The Krebs Cycle
In the Krebs cycle, what type of reaction is responsible for the release of CO2?
a) Decarboxylation
b) Carbonic Acid Neutralization
c) Beta-Oxidation
d) Carbonyl Reduction
A is correct.
In particular steps in the Krebs cycle, carbon dioxide will be generated and released. These reactions involve the removal of a carboxyl group and replacement with a hydrogen atom. This type of chemical reaction is termed decarboxylation. Answer choice B results in the formation of carbonate, CO32-, not CO2. Answer choice C describes the catabolic processing of fatty acids. Answer choice D is incorrect because reduction of a carbonyl would result in the formation of an alcohol, not the release of CO2.
Get 1-on-1 MCAT Tutoring From a Specialist
With MCAT tutoring from MedSchoolCoach, we are committed to help you prepare, excel, and optimize your ideal score on the MCAT exam.
For each student we work with, we learn about their learning style, content knowledge, and goals. We match them with the most suitable tutor and conduct online sessions that make them feel as if they are in the classroom. Each session is recorded, plus with access to whiteboard notes. We focus on high-yield topics if you’re pressed for time. If you have more time or high-score goals, we meticulously cover the entire MCAT syllabus.
Overview of the Krebs Cycle
The Krebs cycle, also known as the citric acid cycle, is a series of chemical processes used by aerobic organisms in order to break down carbon fuels into carbon dioxide, water, and energy. Figure 1 shows a diagram of the eight steps of the Krebs cycle. Overall, the cycle starts with the combination of acetyl-CoA and oxaloacetate and ends with the reformation of oxaloacetate.
As a side note, the starting Krebs cycle reactant, acetyl-CoA, can be supplied by a variety of upstream metabolic pathways, including glycolysis and the subsequent decarboxylation of pyruvate by pyruvate dehydrogeanse complex, as well as the β-oxidation of fatty acids.
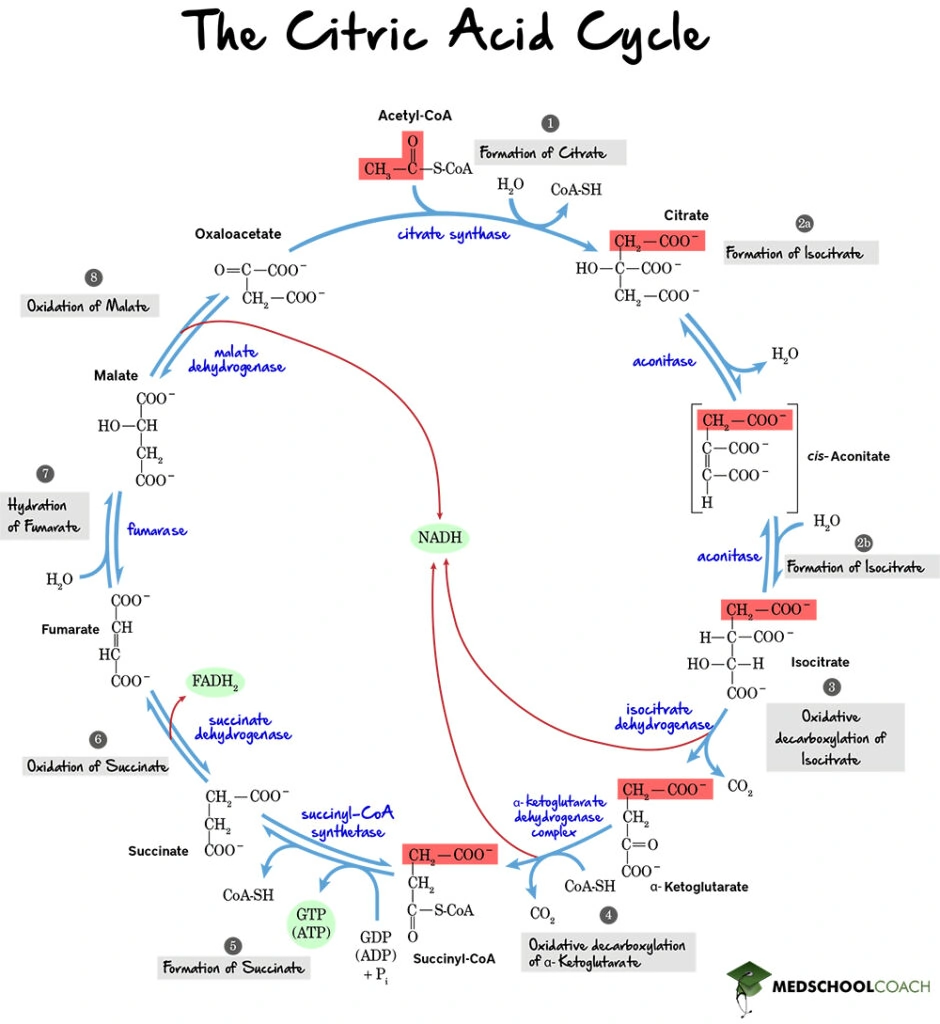
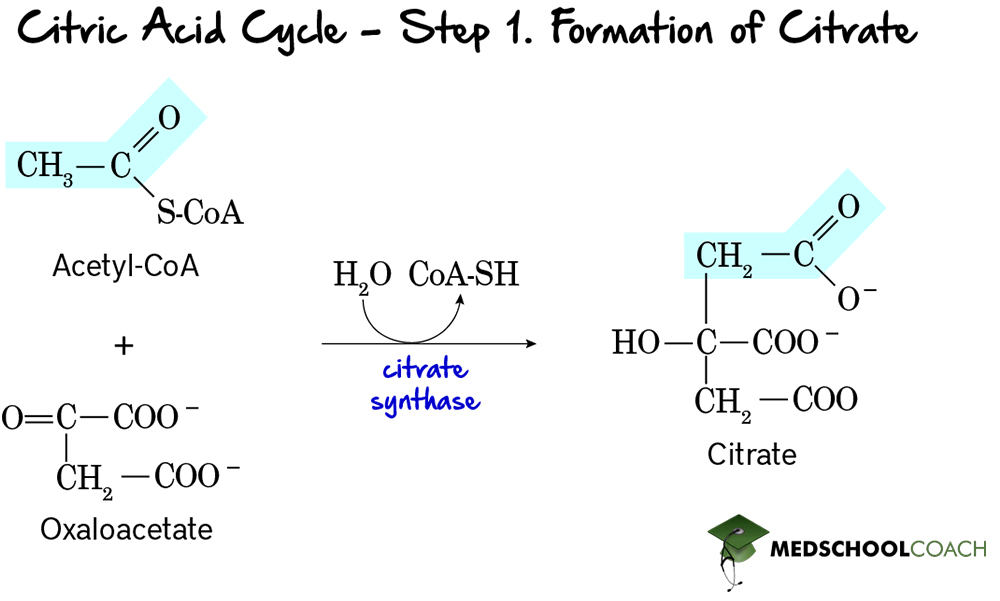
Step 1: Formation of Citrate
The first step of the cycle is the formation of citrate (Figure 2). In this step, acetyl-CoA donates its 2- carbon acetyl group 4-carbon to oxaloacetate, forming 6-carbon citrate. This reaction is catalyzed by the enzyme citrate synthase. Note that anytime a molecule is bound to coenzyme A, it is bound by a thioester bond. Hydrolysis of a thioester bond is highly exergonic and releases a high amount of free energy. This reaction is essential for driving many endergonic or unfavorable processes. In this instance, it is helpful because the oxaloacetate concentration in the mitochondrial matrix is relatively low, and the highly exergonic hydrolysis of the thioester bond helps to push the unfavorable addition reaction forward.
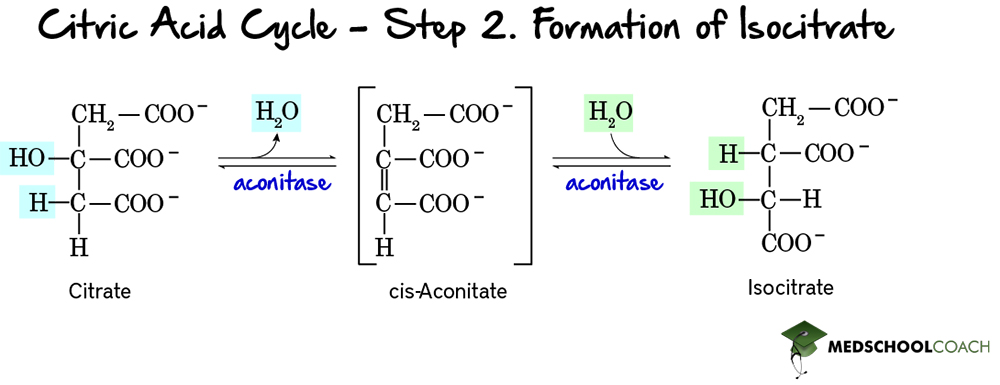
Step 2: Formation of Isocitrate
The second step of the Krebs cycle is the formation of isocitrate (Figure 3). In this two-step reaction. citrate is isomerized to isocitrate. This reaction is catalyzed by aconitase via the formation of an intermediate molecule called cis-aconitate. In the first step of this reaction, a dehydration reaction results in cis-aconitate formation from citrate. The subsequent reaction is a hydration reaction, forming isocitrate from cis-aconitate.
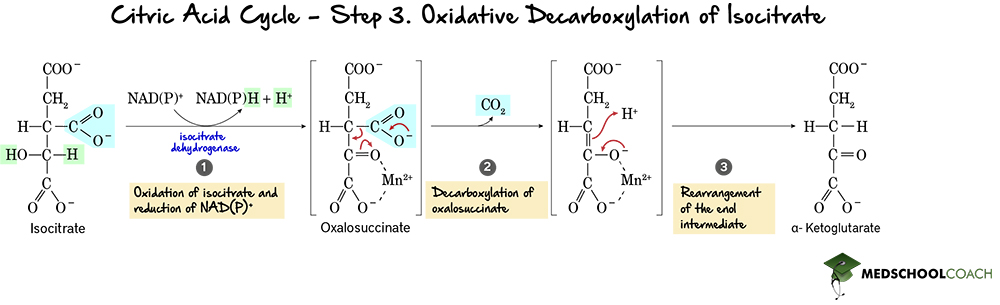
Step 3: Decarboxylation of Isocitrate
The third step in the cycle is the oxidative decarboxylation of isocitrate (Figure 4). Recall that oxidative decarboxylation means that a compound will be oxidized and a CO2 removed to form a new compound. In this case, isocitrate is oxidized and decarboxylated to form α-ketoglutarate and carbon dioxide. Note that one of the carbons from the acetyl group of isocitrate and acetyl-CoA is released as carbon dioxide, making α-ketoglutarate a 5-carbon molecule. Also, as isocitrate is oxidized, NAD+ is reduced to NADH. The NADH will be shuttled to the electron transport chain to donate the energy has captured towards ATP production. This reaction is catalyzed by isocitrate dehydrogenase.
Step 4: Decarboxylation of α-Ketoglutarate
The fourth step involves another oxidation-decarboxylation reaction. This time, α-ketoglutarate is oxidized and decarboxylated to form four-carbon succinyl-CoA and carbon dioxide, and NAD+ is reduced to NADH (Figure 5). Also, note the addition of CoA-SH to form succinyl-CoA. This entire process is catalyzed by the α-ketoglutarate dehydrogenase complex.
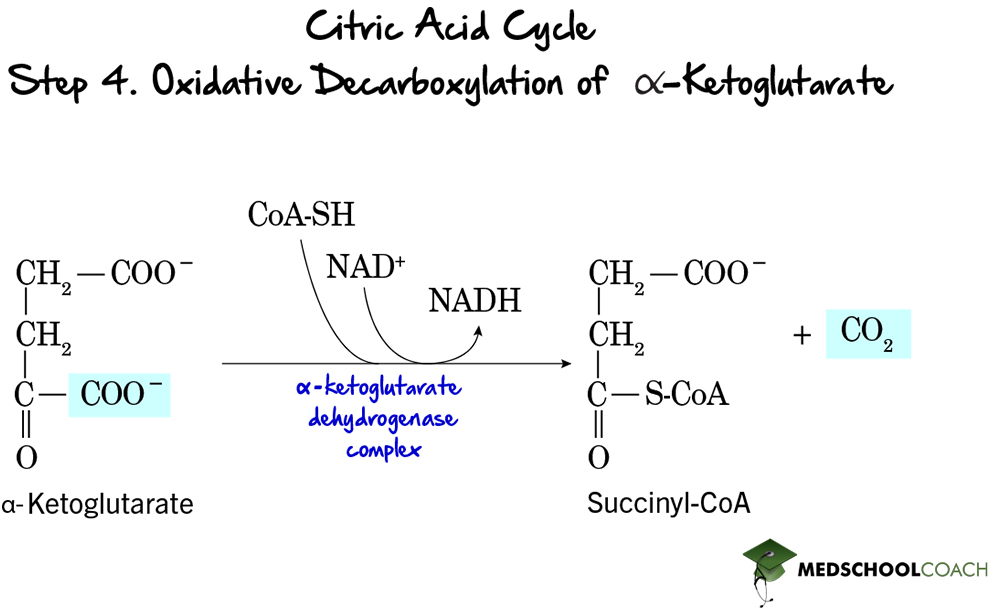
Step 5: Formation of Succinate
At this point in the cycle, two carbons have been removed from acetyl-CoA as carbon dioxide, thus essentially completing the oxidation of glucose. Therefore, steps 5-8 essentially serve to regenerate oxaloacetate so that another cycle can take place. In the fifth step of the Krebs cycle, succinate is formed by hydrolyzing the thioester bond of succinyl-CoA (Figure 6). Remember that the breaking of a thioester bond is highly exergonic can be coupled to an endergonic reaction. In this step, the endergonic reaction performed is the formation of GTP from GDP by substrate-level phosphorylation. This entire reaction is catalyzed by the enzyme succinyl-CoA synthetase.
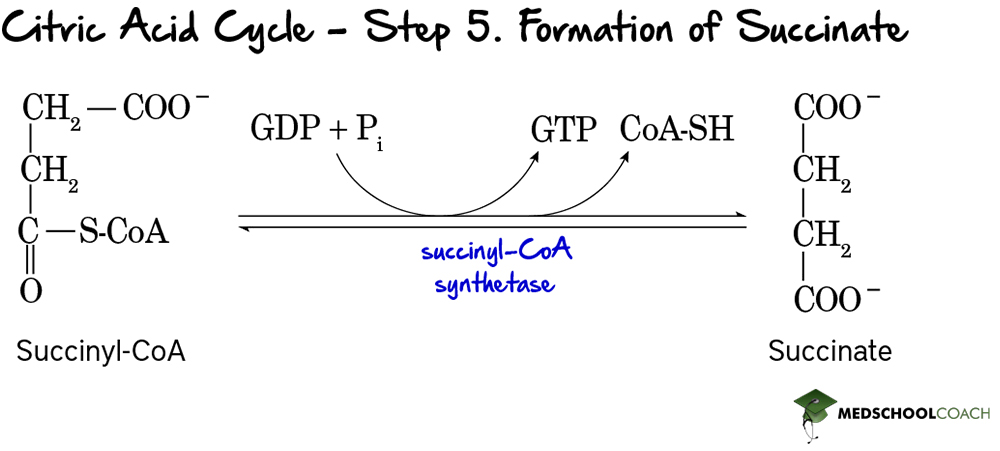
Step 6: Oxidation of Succinate
In the sixth step of the Krebs cycle, succinate is oxidized to fumarate (Figure 7) and FAD is reduced to FADH2. This reaction is catalyzed by the enzyme succinate dehydrogenase. One important thing to note is that succinate dehydrogenase is the only enzyme of the Krebs cycle that participates in both the Krebs cycle and the electron transport chain. In the electron transport chain, succinate dehydrogenase oxidizes FADH2 to FAD and reduces ubiquinone to ubiquinol.
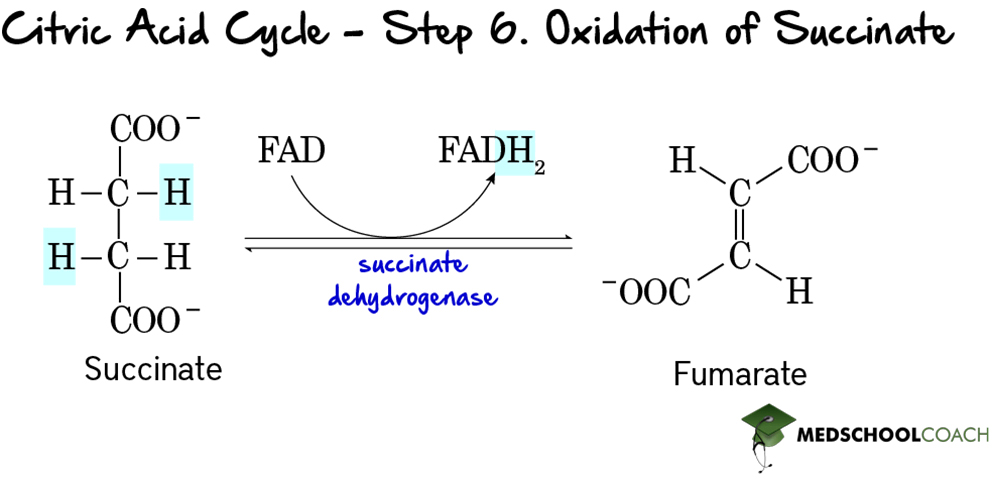
Step 7: Hydration of Fumarate
The seventh step of the cycle is the hydration of fumarate (Figure 8). As previously mentioned, hydration means the addition of water. In this case, water is added to fumarate to form malate in a reaction catalyzed by fumarase. One interesting note is that fumarase is present in many tissues throughout the body, and mutations in this enzyme have been linked to several diseases such as smooth muscle tumors, and even renal cell cancer.
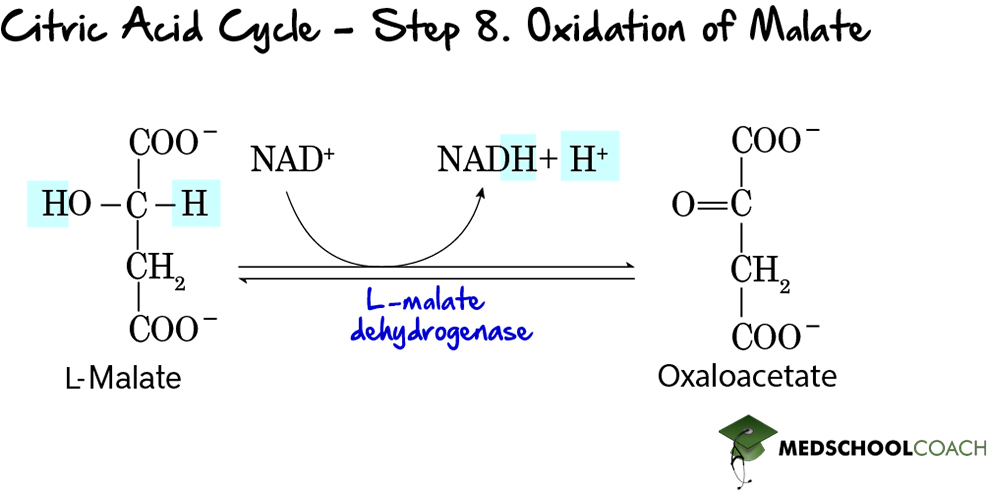
Step 8: Oxidation of Malate
In the last step of the Krebs cycle, malate is oxidized to regenerate oxaloacetate (Figure 9) and a final NAD+ molecule is reduced to NADH. As previously mentioned, the Krebs cycle starts with the generation of oxaloacetate and ends with its regeneration. In this way, as long as acetyl-CoA is present in the body, the Krebs cycle will run continuously.
Explore More MCAT Masterclass Chapters
Take a closer look at our entire MCAT Masterclass or explore our Biochemistry lessons below.

One-on-One Tutoring
Are you ready to take your MCAT performance to a whole new level? Work with our 99th-percentile MCAT tutors to boost your score by 12 points or more!
See if MCAT Tutoring can help me
Talk to our enrollment team about MCAT Tutoring

MCAT Go Audio Course
Engaging audio learning to take your MCAT learning on the go, any time, any where. You'll be on the way to a higher MCAT score no matter where you are. Listen to over 200+ lessons.

MCAT Practice Exams
Practice makes perfect! Our mock exams coupled with thorough explanations and in-depth analytics help students understand exactly where they stand.

MCAT Prep App
Access hundreds of MCAT videos to help you study and raise your exam score. Augment your learning with expert-created flashcards and a question banks.