How are glycolysis and gluconeogenesis regulated differently? This lesson explains the key differences between these processes that you need to know for the MCAT!
Regulation of Glycolysis and Gluconeogenesis
MCAT Biochemistry Chapter 5 - Section 3.1 - Metabolic Pathways - Regulation of Glycolysis and Gluconeogenesis
- Home
- »
- MCAT Masterclass
- »
- Biological and Biochemical Foundations of Living Systems
- »
- Biochemistry
- »
- Regulation of Glycolysis and Gluconeogenesis – MCAT Biochem
Sample MCAT Question
Which of the following is true regarding the hormonal regulation of glucose metabolism?
a) Fructose 2,6-bisphosphate production is activated by glucagon
b) Glucagon inhibits the enzyme fructose 2,6-bisphosphatase
c) Insulin activates the enzyme phosphofructokinase-2
d) Increased insulin increases the glycolytic production of fructose 6-phosphate
C is correct. Insulin activates the enzyme phosphofructokinase-2.
An essential regulator of glycolysis and gluconeogenesis is the intermediate fructose 2,6-bisphosphate. The presence of this metabolite can drive glycolysis and inhibit gluconeogenesis. Its formation is activated by the presence of glucose in the bloodstream and increased insulin. Meanwhile, it is inhibited by decreased insulin and lack of glucose in the bloodstream. The enzyme phosphofructokinase-2, which drives the production of fructose 2,6-bisphosphate from fructose 6-phosphate, is activated by insulin and inhibited by glucagon. Conversely, fructose 2,6-bisphosphate is converted back into fructose 6-phosphate by the enzyme fructose 2,6-bisphosphatase, which is activated by glucagon and inhibited by insulin. Answer choices A and B are the opposite outcomes of glucagon activity. Answer choice D is incorrect, the glycolytic formation of fructose 6-phosphate occurs via a reversible isomerization reaction and occurs in both gluconeogenesis and glycolysis. The regulation of this is not a direct consequence of insulin activity, rather, it is regulated by the concentrations of upstream and downstream F6P intermediates.
Glycolysis and Gluconeogenesis - Reciprocal Regulation
Recall that glycolysis and gluconeogenesis are both pathways of glucose metabolism that produce opposite results. Glycolysis is the breakdown of a molecule of glucose into two molecules of pyruvate and free energy. Gluconeogenesis, on the other hand, is the generation of glucose from two molecules of pyruvate and other substrates. In the body, these two pathways are reciprocally regulated, meaning that situations that activate one pathway also can inhibit the other pathway.
Avoiding a Futile Cycle
This reciprocal regulation is crucial because if both pathways were operating simultaneously in cells, a futile cycle would occur, in which glucose would be broken down to form ATP, which would then be used up to recreate glucose. To demonstrate this point let’s look at the overall net reaction for each pathway.
Glycolysis: glucose + 2 NAD+ + 2 ADP + 2 Pi → 2 pyruvate + 2 NADH + 2 H+ + 2 ATP
Gluconeogenesis: 2 Pyruvate + 4 ATP + 2 GTP + 2 NADH + 2 H+ + 2 H2O → Glucose + 4 ADP + 2 GDP + 6 Pi + 2 NAD+
As shown, if both processes are co-occurring, neither glucose nor pyruvate would increase in concentration, since each is the product of one reaction and the reactant in the other. Further, since gluconeogenesis requires requires 4 ATP and 2 GTP, compared to the 2 ATP produced by glycolysis, running both reactions at once would actually cost energy while simultaneously yielding no result. To avoid this situation, regulation is needed so that when one pathway is running, the other is shut off.
Regulation of Glycolysis and Gluconeogenesis
Metabolic regulation occurs at several key steps of glycolysis and gluconeogenesis. In each of these steps, external factors, such as the concentration of a downstream molecule, can activate or inhibit the enzymes that catalyze the step.
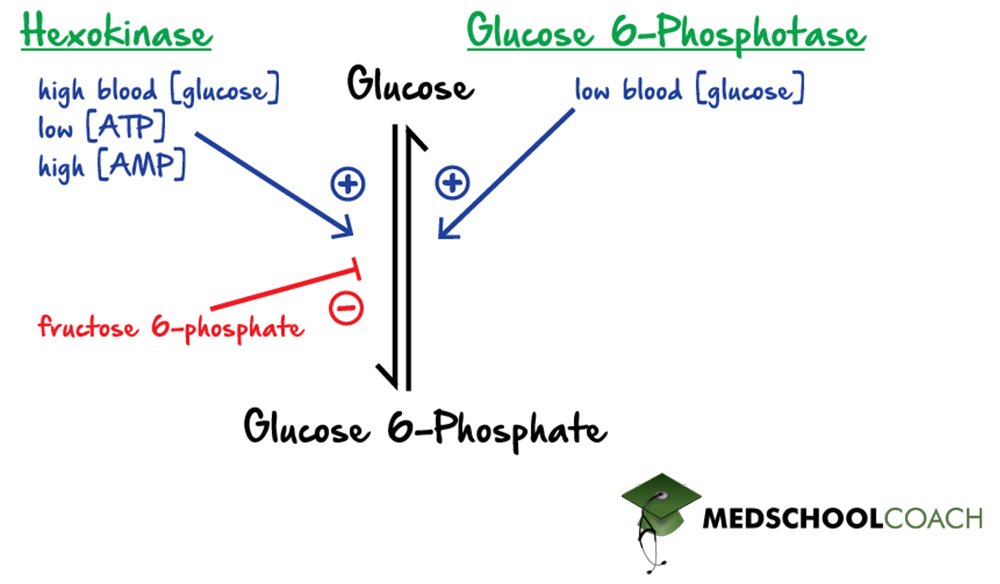
Regulation of Hexokinase and Glucose-6-Phosphotase
The first regulated step of glycolysis (Figure 2) is the creation of glucose 6-phosphate from glucose and ATP. This step is catalyzed by hexokinase. Hexokinase is activated by high blood glucose concentrations, high AMP concentrations, and low cellular ATP concentrations. In other words, if blood glucose concentrations are high, the process of glycolysis is upregulated. Glycolysis is also upregulated when cellular concentrations of ATP are low and concentrations of AMP are high. These two scenarios indicate that the cell is in a low energy state and can directly influence the cell to produce more energy.
Hexokinase can also be inhibited. Specifically, this enzyme is inhibited by fructose 6-phosphate, a downstream intermediate of glycolysis. This type of inhibition is an example of negative feedback. If there are high amounts of fructose 6-phosphate, this likely means that glycolysis has been adequately running and the cell is likely to be in a high energy state. Therefore, a downstream molecule, like fructose 6-phosphate, can inhibit the activity of hexokinase, downregulating glycolysis.
In gluconeogenesis, the reverse reaction, glucose 6-phosphate to glucose, is catalyzed by glucose 6-phosphatase. Glucose 6-phosphatase is activated by low blood glucose concentrations. This should make sense because if blood glucose concentrations are low, the cell will want to upregulate gluconeogenesis to make more glucose.
Regulation of PFK-1 and Fructose-1,6-Bisphosphotase
The next point of regulation in glycolysis is the conversion of fructose 6-phosphate to fructose 1,6-bisphosphate by phosphofructokinase-1 (PFK-1) (Figure 3). Both ADP and AMP are indicators of a low-energy state and therefore activate PFK-1. Recall again that when cells are in a low-energy state, they want to activate glycolysis in order to produce ATP. Similarly, indicators of a high energy state, namely ATP and citrate, inhibit PFK-1, and thereby glycolysis. Citrate is an intermediate of the Krebs cycle (also known as the citric acid cycle). If there is a high concentration of citrate, this indicates that the downstream citric acid cycle is running adequately, and the cell is likely in an energetically satisfied state, meaning glycolysis can be downregulated.
In gluconeogenesis, by contrast, where fructose 1,6-bisphosphate is converted to fructose 6-phosphate, AMP, an indicator of a low-energy state, inhibits the reaction enzyme, fructose 1,6-bisphosphatase. This should make sense because when a cell is in a low energy state, it should not want to produce glucose, but instead break it down to produce energy.
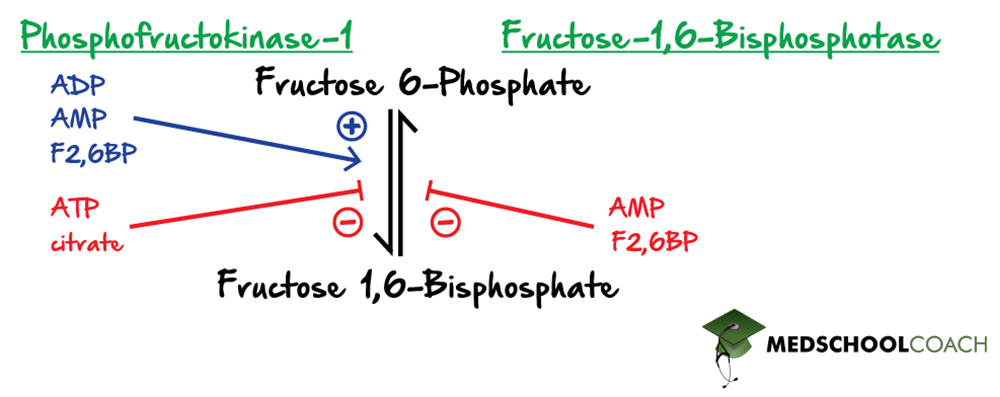
Another critical regulator of this pair of reactions is fructose 2,6-bisphosphate. Note that this molecule is not the same as fructose 1,6-bisphosphate, which is an intermediate of glycolysis and gluconeogenesis. However, fructose 2,6-bisphosphate is an important factor when considering the previously mentioned steps because it mediates the effects of insulin and glucagon on glycolysis and gluconeogenesis. Specifically, fructose 2,6-bisphosphate interacts with PFK-1, activating it in order to upregulate glycolysis. Fructose 2,6-bisphosphate is formed from fructose 6-phosphate and ATP in a reaction catalyzed by phosphofructokinase-2 (PFK-2). PFK-2 itself is regulated by insulin and glucagon. Recall that insulin is released when blood glucose concentrations are high, serving to increase the cellular uptake of glucose. This uptake of glucose by the cells will stimulate glycolysis and inhibit gluconeogenesis. Moreover, insulin also directly activates PFK-2, driving the production of fructose 2,6-bisphosphate, thereby indirectly activating PFK-1 and glycolysis.
The reverse reaction is catalyzed by fructose-2,6-bisphosphatase (FBPase-2) and is also subject to regulation. In general terms, anything that will suppress glycolysis and activate gluconeogenesis will activate FBPase-2. For example, increased glucagon is released when blood glucose concentrations are low. In this state, glycolysis will not proceed. The blood glucagon will activate FBPase-2, which in turn drives the gluconeogenic reaction of fructose 2,6-bisphosphate to fructose 6-phosphate. Fructose 6-phosphate, as previously mentioned, can further inhibit glycolysis and drive gluconeogenesis.
In summary, insulin activates PFK-2 and inhibits FBPase-2, and glucagon activates FBPase-2 and inhibits PFK-2.

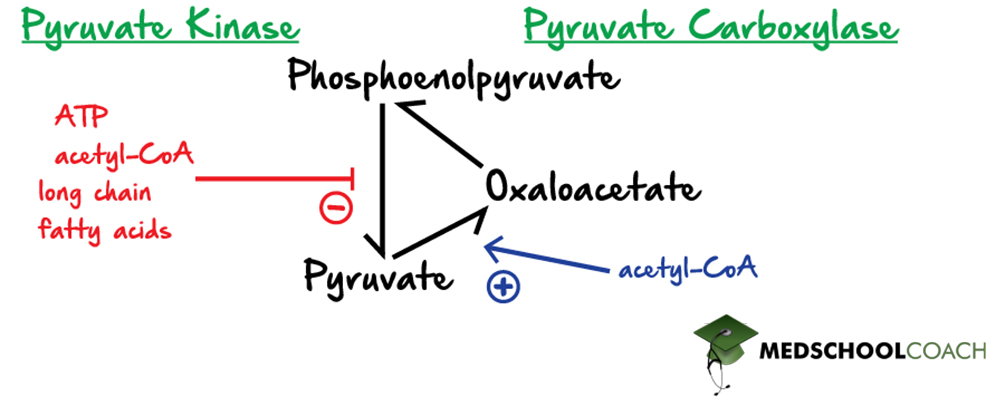
Regulation of Pyruvate Kinase and Pyruvate Carboxylase
The last point of regulation in glycolysis is the conversion of phosphoenolpyruvate to pyruvate by the enzyme pyruvate kinase. In gluconeogenesis, the reverse conversion of pyruvate to phosphoenolpyruvate, requires two enzymes and also involves the intermediate production of oxaloacetate. Specifically, pyruvate carboxylase catalyzes the first step of converting pyruvate to oxaloacetate. Oxaloacetate is then decarboxylated and phosphorylated to form phosphoenolpyruvate with the help of the enzyme phosphoenolpyruvate carboxykinase. Again, if there are indications that the cell is in a high-energy state, such as increased concentrations of ATP, acetyl-CoA, or long-chain fatty acids, then glycolysis will be inhibited. Recall that acetyl-CoA holds energy, if the cell does not require energy production, this energy must be stored. The best way to store such energy is to convert acetyl-CoA to glucose to be stored in glycogen. In this way, acetyl-CoA will activate pyruvate carboxylase, which will upregulate gluconeogenesis.
Explore More MCAT Masterclass Chapters
Take a closer look at our entire MCAT Masterclass or explore our Biochemistry lessons below.

One-on-One Tutoring
Are you ready to take your MCAT performance to a whole new level? Work with our 99th-percentile MCAT tutors to boost your score by 12 points or more!
See if MCAT Tutoring can help me
Talk to our enrollment team about MCAT Tutoring

MCAT Go Audio Course
Engaging audio learning to take your MCAT learning on the go, any time, any where. You'll be on the way to a higher MCAT score no matter where you are. Listen to over 200+ lessons.

MCAT Practice Exams
Practice makes perfect! Our mock exams coupled with thorough explanations and in-depth analytics help students understand exactly where they stand.

MCAT Prep App
Access hundreds of MCAT videos to help you study and raise your exam score. Augment your learning with expert-created flashcards and a question banks.